 209, indicates the mass number of the longest-lived isotope of the element. moles Cu/no, moles O) in the sample?: 1 What is the formula of the oxide? (The atom ratio equals the mole ratio, and is expressed using the smallest integers possible. See also a copy of the periodic table with atomic weights to five significant. moles = mass Cu/molar mass Cu) moles How many grams of O are there in the sample? (The mass of the sample mass of Cu plus the mass of O.) g How many moles of O are there in the sample? Moles What is the mole ratio (no. This table can be obtained in PDF format here. This black and white periodic table contains the accepted atomic weights of each element as accepted by the IUPAC. According to the periodic table, 1 mol of U has a mass of 238.03 g, so the mass of 2 mol is twice that. This periodic table contains the atomic masses of the elements as accepted by the IUPAC (09-2013). The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. How many moles of Cu are there in the sample? (No. One mole of Al atoms has a mass in grams that is numerically equivalent to the atomic mass of aluminum. In a certain compound of copper and oxygen, Cu_x O_y, we find that a sample weighing 0.6349 g contains 0.5072 g Cu. Find the molar mass of Cu g Cl g H g O g H_2O g If one can find the ratio of the number of moles of the elements in a compound to one another, one can find the formula of the compound. For a compound substance, the molar mass is equal to the mass in grams that is numerically equal to the sum of the atomic masses in the formula of the substance. By way of example, the first aspect, T, may be the season that it was discovered. 
The molar mass of an element is simply the mass in grams of that element that is numerically equal to its atomic mass. Thermal Conductivity0.1805 W/ (m K) Specific Heat 14300 J/ (kg K) Heat Fusion 0.558 kJ/mol. Printable Periodic Table With Molar Mass The Routine Dinner table is actually a clinical graph or chart that organizations components by their period of development. 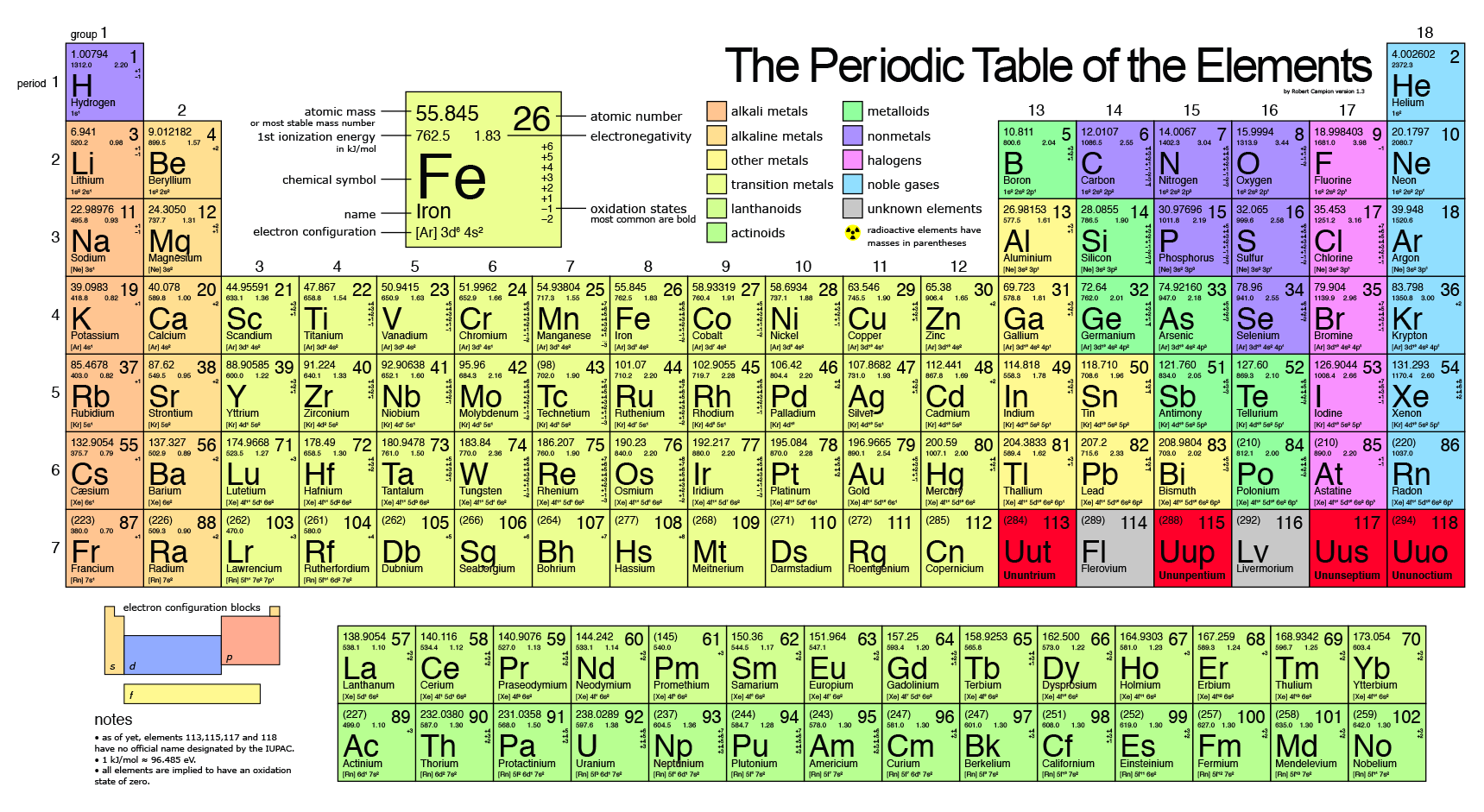
To find the mass of a mole of an element, one looks up the atomic mass of the element in a table of atomic masses (see Appendix III or the periodic Table). The Periodic Table of the Elements Average relative masses are 2001 values, rounded to two decimal places. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
